Science and Medicine

Jeyou Pharma specializes in the research, development, manufacturing, and commercialization of innovative medicines. Focusing on a “3+1” indication matrix—including oncology, nephrology, pain, and autoimmune diseases—the company is committed to providing patients worldwide with medicines that offer unique clinical value and address unmet medical needs. Jeyou Pharma has established an end-to-end R&D system covering project selection and initiation, innovative drug discovery, preclinical pharmacology and toxicology studies, clinical trials, regulatory filing and market launch, as well as comprehensive project management, thereby building a one-stop R&D capability spanning from drug discovery to regulatory submission and market launch.







Jeyou Pharma integrates molecular design, clinical pathway design, CMC processes, and regulatory strategies into a unified project management framework, ensuring a seamless transition from the laboratory to clinical trials. The company has established a data-driven, full-lifecycle management system to enable real-time monitoring of the R&D process and optimize resource allocation. Additionally, Jeyou Pharma has forged partnerships with leading global research institutions and multinational pharmaceutical companies to enhance its technological capabilities and has assembled a team of external expert advisors to provide scientific support for major decision-making.

Jeyou Pharma’s core team of scientists each has over 15 years of experience in drug discovery and development, including experience at large multinational pharmaceutical companies. They have led or participated in the development of numerous new drugs that are now on the market or in clinical trials, and have achieved significant success in the development of innovative drugs.

Through iterative development using an AI platform, Jiyu Pharmaceutical has established five R&D platforms that feature exceptionally high competitive barriers and efficiency.
JY
JY
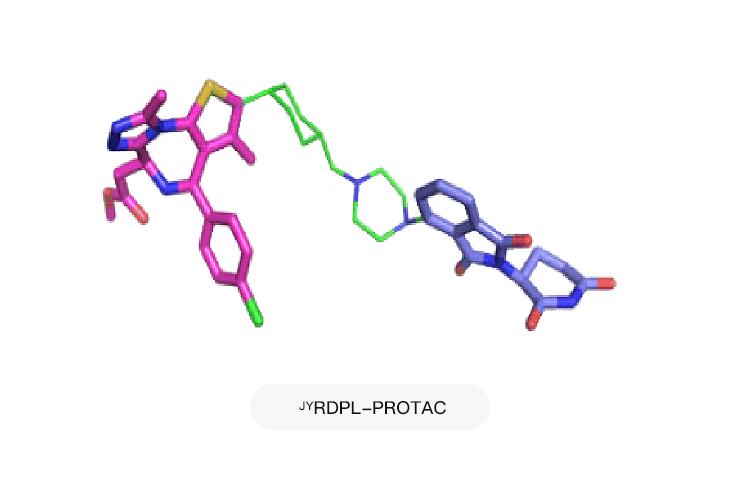
RDPL-PROTAC: A PROTAC platform for rapid molecular design based on proprietary ligands and computational methods
RDPL-PROTAC: A PROTAC platform for rapid molecular design based on proprietary ligands and computational methods
Project Applications: JMKX002992,JMN-2505,JMN-2506
Using platform technology, PROTAC molecules with high biological activity and favorable pharmacokinetics can be efficiently designed for degradation targets.
Maturation and storage of the proprietary E3 ligand
AIDD's Computationally Enabled Multi-Dimensional Capabilities for Efficient Advancement
JY
JY
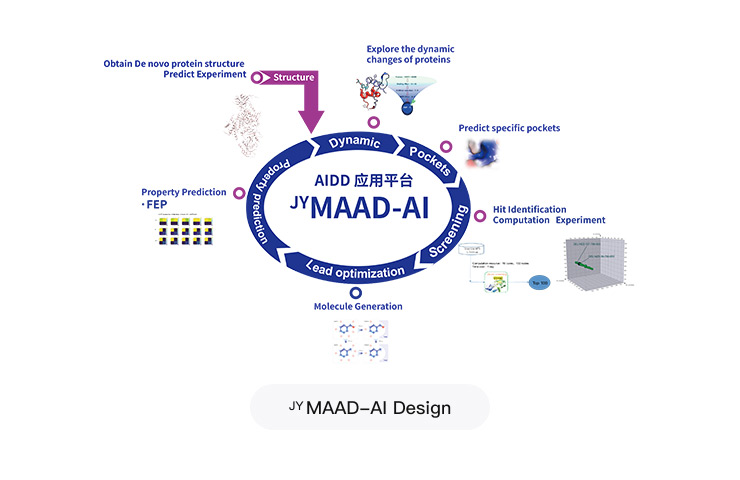
MAAD-AI Design Integrated Algorithm-Driven AI-Powered Drug Discovery Platform
MAAD-AI Design Integrated Algorithm-Driven AI-Powered Drug Discovery Platform
Project Applications: ~30 targets and projects
In-house development combined with external partnerships, with comprehensive software and hardware deployment of the latest algorithms and ample GPU computing power
Over 20 software tools, including industry-leading AI capabilities such as structure prediction, dynamics simulation, molecular generation, and FEP calculations
Empowering molecular design, accelerating the FF project, and launching the FIC project
JY
JY
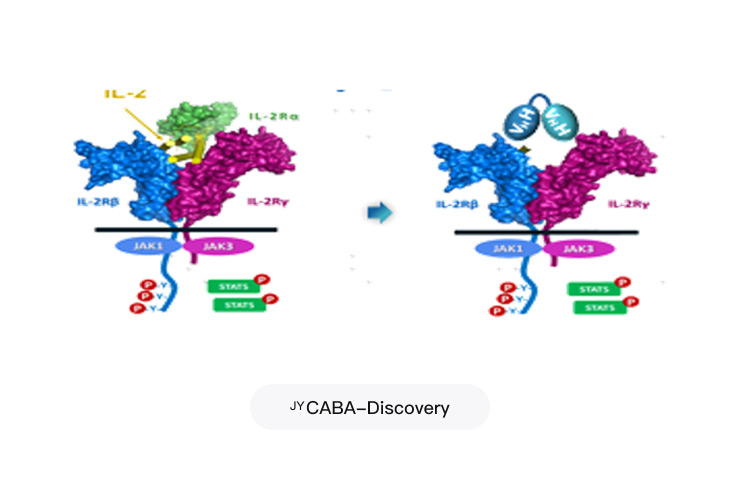
CABA-Discovery Cytokine-Activated Bispecific Antibody Discovery Platform
CABA-Discovery Cytokine-Activated Bispecific Antibody Discovery Platform
Project Applications: 2401 bispecific antibody, 2403, and other innovative trispecific antibody projects
Analysis of millions of antibody sequences
Increased the throughput of antibody sequence screening by a factor of thousands
Identify novel epitopes that cannot be identified using many traditional methods
JY
JY
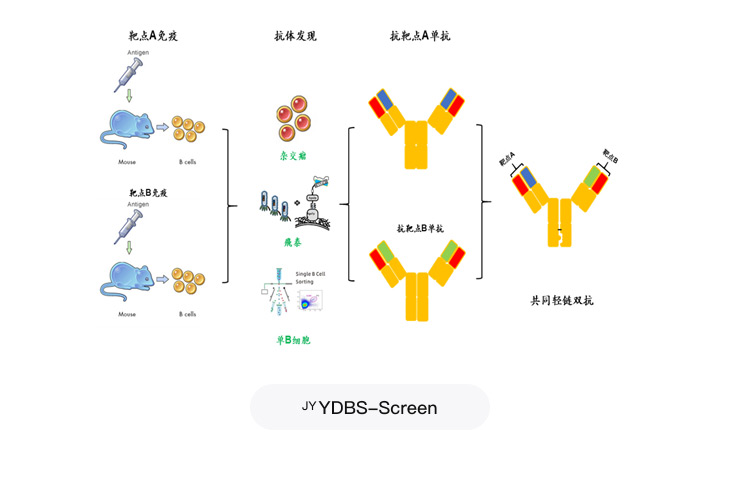
YDBS-Screen Yeast-Based Light Chain Dual-Antibody Screening Platform
YDBS-Screen Yeast-Based Light Chain Dual-Antibody Screening Platform
Project Applications: 2005, 2202, and other light-chain-targeted bispecific antibody projects
A dual-antibody screening and optimization platform for yeast surfaces
An Innovative Yeast Display Platform Based on IgG Formats
Scaling up antibody sequence discovery to millions of sequences per run
JY
JY
Pepfold–DRP A platform for the precise folding of polycyclic peptides via disulfide bonds
Pepfold–DRP A platform for the precise folding of polycyclic peptides via disulfide bonds
Project Application: JMKX2301, trans-brain peptide conjugate
A large-scale library of 109-mer peptides and a unique library of polycyclic peptides containing multiple disulfide bonds
Significantly improve the success rate of producing correctly folded multipeptide chains

Over 40 projects currently in development:including over 30 small-molecule projects and over 10 large-molecule projects, with 9 in Phase II clinical trials and 10 in Phase I clinical trials
| Featured Products | Mechanism/Target | Indications | PCC IND PH1 PH2 PH3 | Partner |
|---|---|---|---|---|
|
JMKX003142 |
V2R | ADPKD |
|
|
| Renal edema |
|
|||
|
JMKX003142-IV |
Cardiac edema |
|
||
|
JMKX003002 |
NHE3 | Hyperphosphatemia (in hemodialysis patients) |
|
|
|
JMB-2201 |
- |
|
||
|
JMKX001899 |
KRAS-G12C | 2L NSCLC |
Key Clinical Trials |

|
|
JMKX000197 |
STING | Malignant pleural effusion |
|
|
|
JMB-2301 |
- |
|
||
|
JMKX000189 |
S1P1 | CIPN |
|
|
| UC |
|
|||
|
JYB1904 |
lgE | Asthma |
|

|
| Chronic spontaneous urticaria |
|
|||
| Chronic rhinitis |
|
|||
|
JMKX003948 |
Hif2a | wAMD |
|
|
|
JMKX002992 |
AR | Tumor |
|

|


Jeyou Pharmaceuticals has established a comprehensive pharmacovigilance system to manage the safety of its products throughout their entire lifecycle. Team members possess strong medical backgrounds and extensive experience in the pharmacovigilance industry, ensuring that all pharmacovigilance activities fully comply with the requirements of relevant regulatory authorities. Based on ICH technical guidelines, the Pharmacovigilance Department has developed sustainable improvement protocols centered on safety information monitoring, evaluation, and risk management. Through continuous oversight of the implementation of the quality policy, the department maintains and enhances the compliant operation of the pharmacovigilance system. By fully fulfilling its responsibilities for safety information monitoring, identification, evaluation, and risk management, the Pharmacovigilance Department continuously refines the safety profiles of the company’s products while minimizing product safety risks to the greatest extent possible. This ensures a favorable benefit-risk balance to protect and promote public health.
If a patient experiences a suspected adverse event, please consult a healthcare professional. To report an adverse event to us, please contact jyadr@jeyoupharma.com.