Previous
Jeyou Pharma's innovative monoclonal antibody JYB1904 Receives Approval for Clinical Trials in a New Indication
Shanghai Jeyou Pharmaceutical Technology Co., Ltd. (hereinafter referred to as “Jeyou Pharmaceutical”) announced that two Class 1 novel biopharmaceuticals developed by its Institute of Innovative Macromolecular Drugs have recently been approved to proceed with clinical trials: JMB2004 Injection, China’s first-in-class recombinant fully human monoclonal antibody for the treatment of sepsis, has been approved by the U.S. Food and Drug Administration (FDA) to conduct clinical trials in the United States for the treatment of sepsis and septic shock; JYB1931 Injection, an innovative recombinant humanized MASP-2 monoclonal antibody, has been approved by the National Medical Products Administration (NMPA) to conduct domestic clinical trials for patients with IgA nephropathy.
JMB2004 Injection is the first domestically developed antibody drug for the treatment of sepsis
The U.S. clinical trial application for JMB2004 Injection was approved by the FDA on December 19, 2023, authorizing the conduct of clinical trials for the treatment of sepsis and septic shock. Globally, there are over 48.9 million new cases of sepsis annually, resulting in 11 million deaths. In China, there are approximately 2.5 million sepsis patients each year, with over 700,000 fatalities. Sepsis is one of the major diseases posing a serious threat to human health. Currently, clinical management primarily relies on non-specific treatments such as anti-infective therapy and symptomatic support; there are no effective targeted therapeutic drugs available.
Preclinical studies have shown that treatment with the JMB2004 antibody can significantly improve sepsis symptoms and increase animal survival rates. Compared to reference antibodies, the JMB2004 antibody exhibits lower immunogenicity and higher affinity, as well as advantages such as the absence of toxic side effects, high safety, and low dosing frequency. Currently, there are no antibody drugs available in China for the treatment of sepsis. As the first domestically developed antibody drug for sepsis, JMB2004 is expected to fill this therapeutic gap and become a Breakthrough Innovation Candidate (BIC) drug.
Preclinical studies show that JYB1931 injection, when combined with MASP-2, offers significant advantages
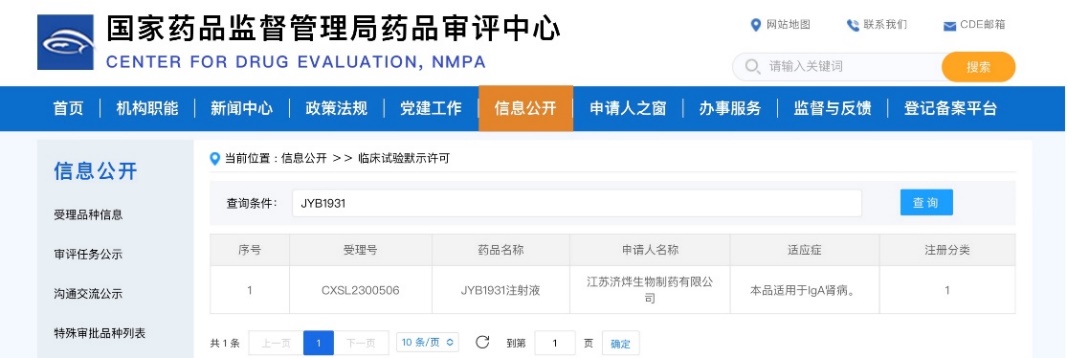
On October 16, 2023, JYB1931 Injection received approval from the NMPA to conduct clinical trials for patients with IgA nephropathy. JYB1931 is an innovative drug—a recombinant humanized monoclonal antibody targeting MASP-2—independently developed by the Jeyou Pharmaceutical Macromolecular Innovation Institute. It holds proprietary intellectual property rights, is classified as a Class 1 new biological product, and is intended for the treatment of IgA nephropathy.
IgA nephropathy is the most common primary glomerular disease in China. It is a severe, life-threatening, progressive immune-mediated disorder that leads to gradual decline in renal function and ultimately results in renal failure. MASP-2 (mannan-binding lectin serine protease 2) is an effector enzyme in the lectin pathway of the complement system. It cleaves C2 and C4, thereby activating the complement system and mediating the progression of inflammatory diseases. Inhibiting MASP-2 can alleviate the progression of various inflammatory diseases or rare diseases.
Preclinical data indicate that JYB1931 binds to MASP-2 with high affinity and specificity, outperforming the most advanced drug of its class currently in clinical development by nearly 100-fold. Furthermore, in human MASP-2 transgenic mice, JYB1931 exhibits a longer half-life and higher peak plasma concentration at the same dose, and it significantly reduces renal cell apoptosis caused by proteinuria.

The new indication is chronic spontaneous urticaria; the previously approved treatment for moderate-to-severe allergic asthma has completed Phase I trials……
2024/09/01
Both drugs are Class 1 new drugs, targeting sepsis and IgA nephropathy, respectively……
2024/08/01
The treatment areas include cancer, kidney disease, and infectious diseases...
2023/25/12