Previous
Jeyou Pharma and RAPT Announce Exclusive Licensing Agreement for a Novel Long-Acting IgE Antibody
Shanghai Jeyou Pharmaceutical Technology Co., Ltd. (hereinafter referred to as “Jeyou Pharmaceutical”) announced that the National Medical Products Administration has approved the clinical trial application for a new indication of JYB1904 Injection, which was independently developed by the company’s Institute of Macromolecular Innovation. The company will now conduct domestic clinical trials for the treatment of chronic spontaneous urticaria.
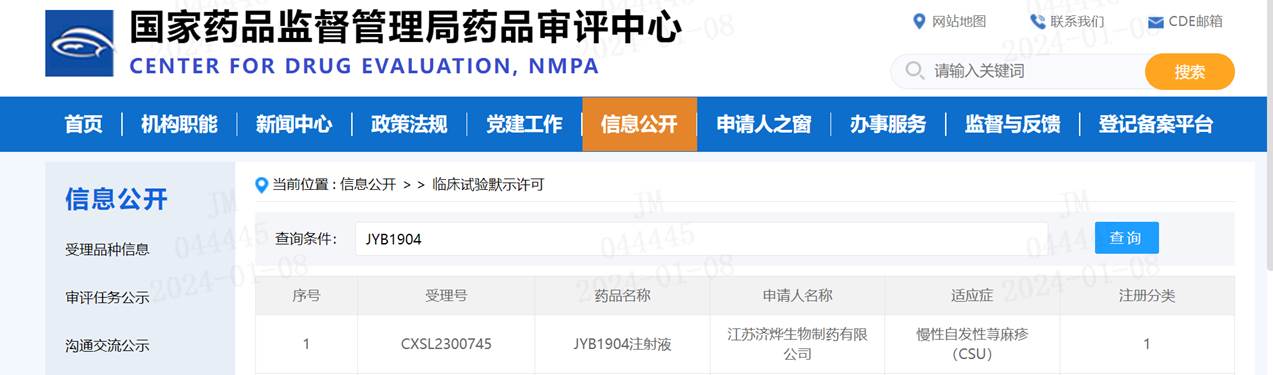
JYB1904 is an innovative, IgE-targeted recombinant humanized monoclonal antibody independently developed by the Jeyou Pharmaceutical Macromolecular Innovation Institute. It holds proprietary intellectual property rights and is classified as a Class 1 new biological product. Previously approved for clinical trials in moderate-to-severe allergic asthma, data from the completed Phase I clinical trial demonstrate good safety and a long half-life, supporting the initiation of Phase II clinical trials. The approval for clinical trials in chronic idiopathic urticaria marks the second clinical indication for JYB1904.
Chronic idiopathic urticaria is a common chronic skin disease primarily characterized by hives and itching. Approximately 20% of patients experience angioedema, which causes significant disruption to their daily lives. Furthermore, nearly half of patients show an inadequate response to second-generation oral H1 antihistamines, creating an urgent need for medications with longer-lasting efficacy to control the disease.
Compared to currently available drugs, JYB1904 exhibits higher affinity, greater humanization, and superior stability, enabling administration as a high-concentration subcutaneous injection. Preclinical study results indicate that JYB1904 can specifically bind to free IgE, blocking the binding of IgE to its receptor, with both in vitro biological activity and in vivo efficacy superior to current drugs. Furthermore, both in vivo animal studies and Phase I clinical data demonstrate that JYB1904 possesses a significantly prolonged half-life.
These findings indicate that JYB1904 injection holds excellent clinical therapeutic potential. It may offer a new treatment option for allergic conditions such as moderate-to-severe allergic asthma and chronic idiopathic urticaria, thereby improving patients’ quality of life and alleviating their disease burden. Jeyou Pharmaceuticals will actively advance the clinical trials of JYB1904 and expand its clinical application in the treatment of allergic diseases such as chronic spontaneous urticaria, striving to benefit more patients.

The new indication is chronic spontaneous urticaria; the previously approved treatment for moderate-to-severe allergic asthma has completed Phase I trials……
2024/09/01
Both drugs are Class 1 new drugs, targeting sepsis and IgA nephropathy, respectively……
2024/08/01
The treatment areas include cancer, kidney disease, and infectious diseases...
2023/25/12