Previous
Jeyou Pharma's Two Innovative Macromolecular Drugs Approved for Clinical Trials
Shanghai Jeyou Pharmaceutical Technology Co., Ltd. announced that six Class 1 new chemical entity (NCE) drug candidates from its Small Molecule Innovation Institute have recently received consecutive approvals to proceed with clinical trials. The six approved innovative small-molecule drugs target therapeutic areas including cancer, kidney disease, and anti-infective diseases. Of these, four have been approved by the National Medical Products Administration (NMPA) to conduct clinical trials in China, while two have been approved by the U.S. Food and Drug Administration (FDA) to conduct clinical trials in the United States.
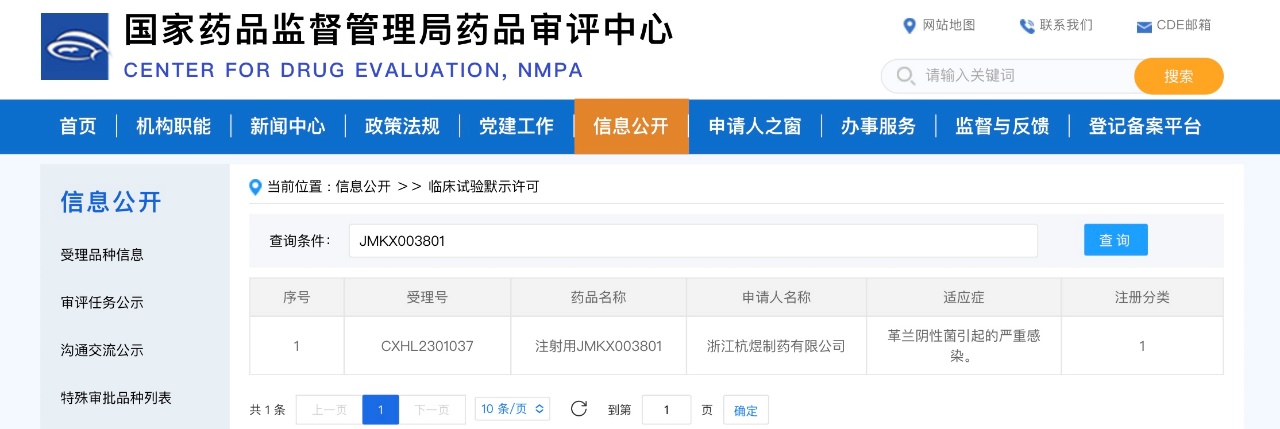
On December 20, JMKX003801 Injection was approved by the National Medical Products Administration, which authorized the conduct of clinical trials for the treatment of severe infections caused by Gram-negative bacteria. The incidence of antibiotic resistance, particularly to carbapenems, in Escherichia coli, Klebsiella pneumoniae, Acinetobacter baumannii, and Pseudomonas aeruginosa has been rising year by year. Mechanistically, JMKX003801 is well-positioned to overcome the resistance seen with existing carbapenems. Preclinical studies indicate that it possesses a broad spectrum of antimicrobial activity, and it is expected to address the current clinical challenges posed by antibiotic resistance.
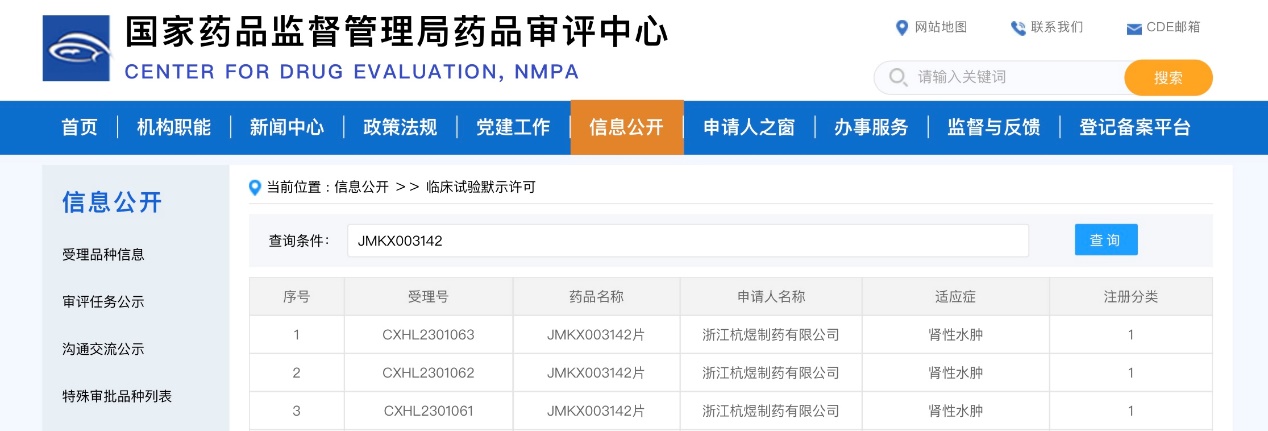
On December 12, the National Medical Products Administration approved JMKX003142 for clinical trials to treat renal edema. The total number of people with chronic kidney disease (CKD) in China is estimated to be as high as 120 million, and in the later stages of CKD, increased proteinuria can lead to edema. This drug is a latest-generation diuretic; research data indicates that it promotes diuresis while causing fewer side effects, and it is expected to improve the quality of life for patients with kidney disease.
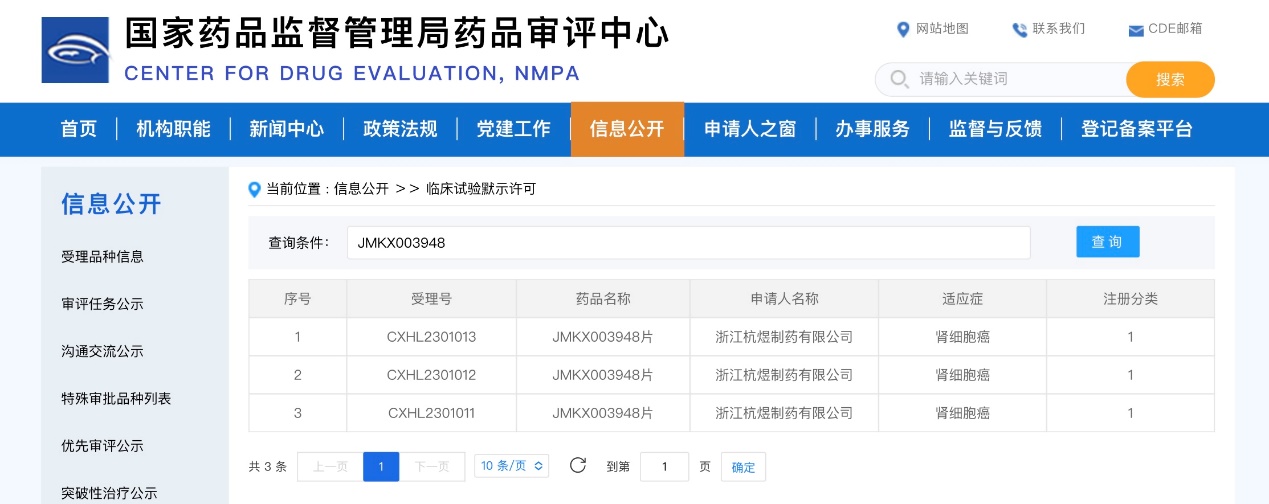
On November 28, JMKX003948 tablets were approved by the National Medical Products Administration (NMPA) to proceed with clinical trials for renal cell carcinoma. There are 60,000 to 80,000 new cases of kidney cancer in China each year. Currently, there is a lack of drugs with novel mechanisms of action for patients who have failed treatment with TKIs and/or PD-1 immunotherapy. JMKX003948, through its distinct therapeutic mechanism and pathways, has demonstrated significant efficacy in preclinical studies in animal models, both as a monotherapy and in combination therapy.

On December 12, the U.S. clinical trial application for JMKX000197 injection was approved by the FDA. This innovative drug had previously been approved by the National Medical Products Administration (NMPA) on October 19, authorizing the conduct of clinical trials for non-muscle-invasive bladder cancer in patients who have failed BCG therapy. Bladder cancer is the most common malignant tumor of the urinary system, with approximately 80,000 new cases diagnosed annually in China. JMKX000197 modulates the immune system and cytokines. Preclinical studies have demonstrated significant antitumor activity in animal models that did not respond to BCG therapy.
Earlier, on November 9, the U.S. clinical trial application for JMKX003002 tablets was also approved by the FDA, authorizing clinical trials for the treatment of hyperphosphatemia in patients with end-stage renal disease undergoing dialysis. The total number of chronic kidney disease (CKD) patients in China is estimated to be as high as 120 million. CKD can lead to hyperphosphatemia and associated mineral and bone disorders (CKD-MBD). This drug is a latest-generation phosphate binder with good oral compliance. Compared to competing products, it exhibits higher in vitro persistence, offering the potential for sustained reduction of serum phosphorus levels and improved quality of life for kidney disease patients.

The new indication is chronic spontaneous urticaria; the previously approved treatment for moderate-to-severe allergic asthma has completed Phase I trials……
2024/09/01
Both drugs are Class 1 new drugs, targeting sepsis and IgA nephropathy, respectively……
2024/08/01
The treatment areas include cancer, kidney disease, and infectious diseases...
2023/25/12