Previous
Validating a Comprehensive R&D System and Pioneering New Frontiers in Combination Therapies | Jeyou Pharma’s First Class 1 Innovative Drug, Jielemei®, Approved for Market Launch
[Abstract] Global pharmaceutical giant GSK has announced the acquisition of RAPT Therapeutics, the U.S. partner of Jeyou Pharmaceutical, for approximately $2.2 billion. The core of the deal centers on JYB1904, an innovative macromolecular drug independently developed by Jeyou Pharmaceutical. Following the interest shown by Roche in its small-molecule pipeline candidate JMKX002992, Jeyou Pharma has now secured substantial backing from GSK in the large-molecule sector. This marks Jeyou as one of the few domestic innovative drug companies capable of delivering “global-class” products in both the small-molecule and large-molecule fields.
On January 20, GlaxoSmithKline (GSK) announced it had reached a definitive agreement to acquire Jeyou Pharma’s U.S. partner, RAPT Therapeutics (“RAPT”) (NASDAQ: RAPT), for $2.2 billion. RAPT is a California-based clinical-stage biopharmaceutical company dedicated to developing new therapies for patients with inflammatory and immune-mediated diseases. The acquisition includes Ozureprubart (JYB1904), a long-acting anti-immunoglobulin E (IgE) monoclonal antibody currently in Phase IIb clinical development for the prevention of food allergies. (Related in-depth industry analysis):https://mp.weixin.qq.com/s/PWlXvK99iylOjA8_cgoqbA)
IgE is a clinically validated target and the only approved systemic therapy proven to protect patients from harmful allergic and inflammatory immune responses. Approximately 94% of severe food allergies are caused by IgE-mediated reactions.
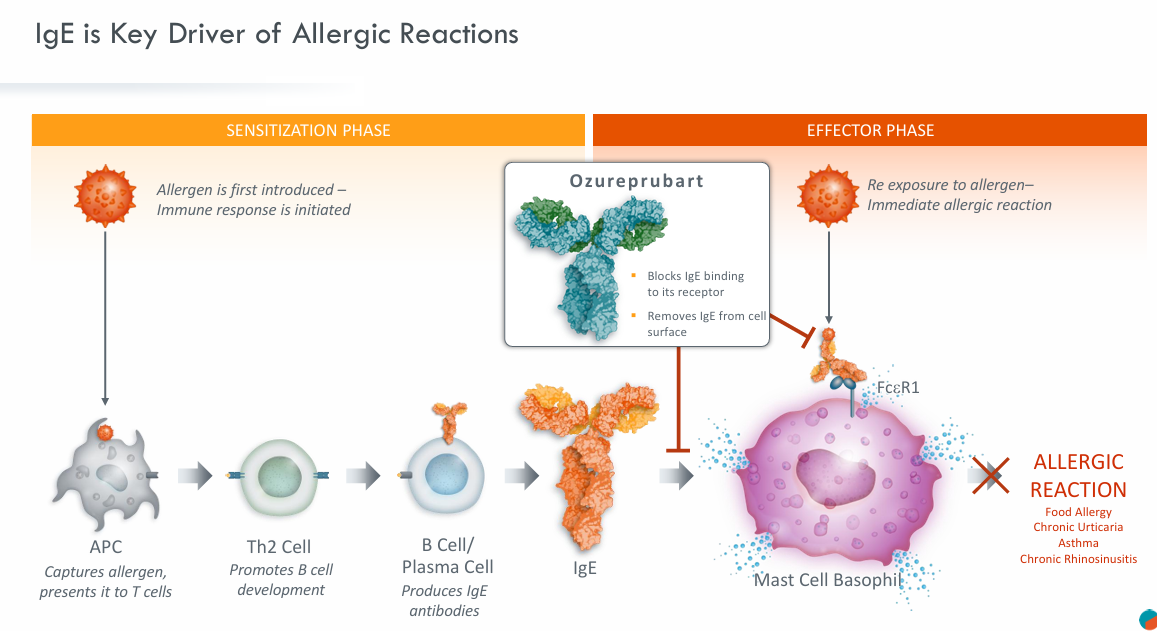
Data from the Phase IIb trial (pretigE) evaluating Ozureprubart (JYB1904) as a monotherapy are expected to be released in 2027, and the Phase III trial will focus on high-risk adult and pediatric populations. In the United States, more than 17 million people have been diagnosed with food allergies, and over 1.3 million experience severe reactions. This results in more than 3 million patient visits and emergency room visits each year.
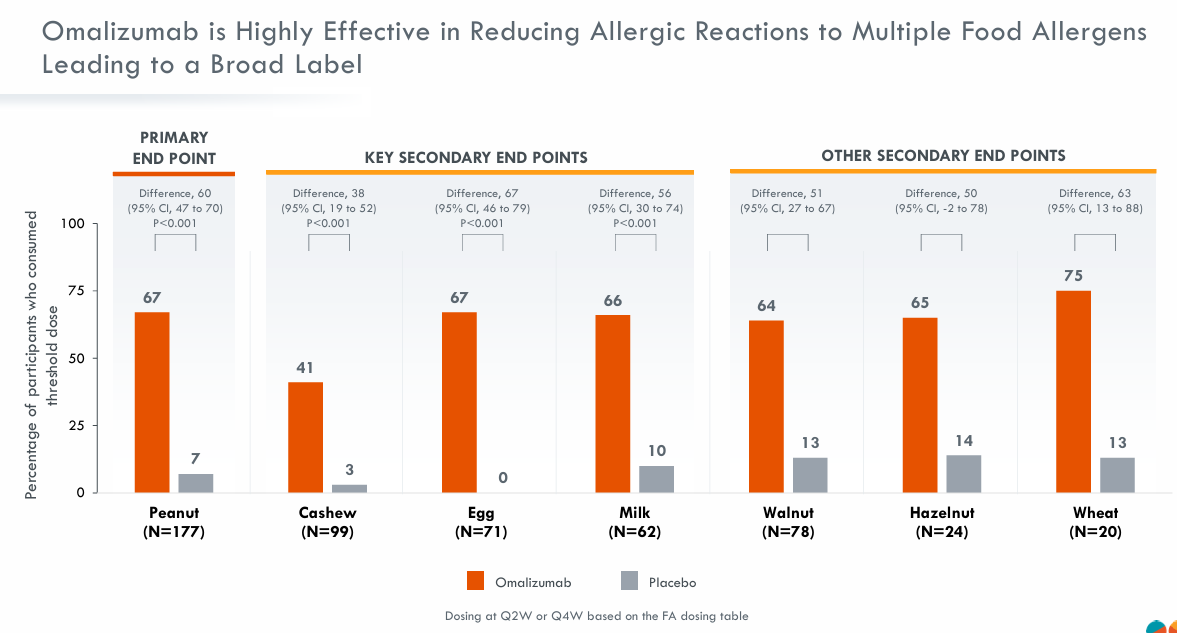
Tony Wood, Chief Scientific Officer at GlaxoSmithKline, said: “The addition of Ozureprubart (JYB1904) brings another promising new, potentially best-in-class treatment to GlaxoSmithKline’s pipeline. Food allergies can have serious health implications for patients on current treatments, which require injections every two weeks. Ozureprubart (JYB1904) offers the opportunity for sustained protection with dosing every 12 weeks, which aligns with our approach to acquiring assets designed to address validated targets and meet significant unmet medical needs.”
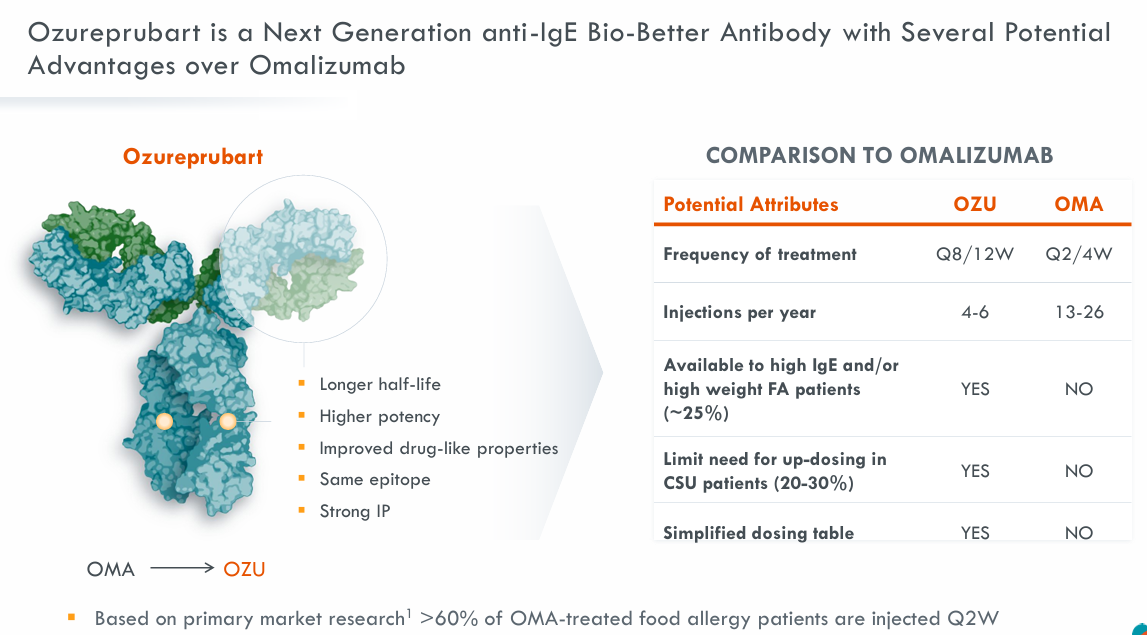
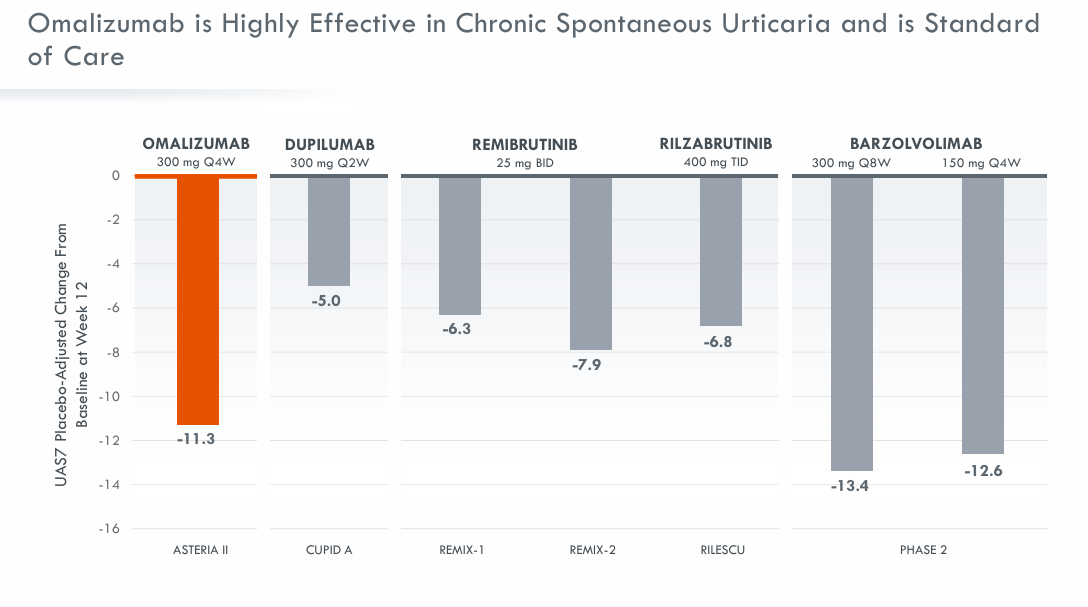
Brian Wong, President and CEO of RAPT Therapeutics, said: “ “We are pleased to enter into this agreement with GlaxoSmithKline, which provides an attractive path forward for our programs, particularly the opportunities we envision for Ozureprubart (JYB1904) in food allergies. This transaction has the potential to leverage GlaxoSmithKline’s global development and commercialization capabilities, resources, and infrastructure, ultimately delivering value to our pipeline, patients, and shareholders.”
Under the terms of the agreement, GlaxoSmithKline has acquired global rights to Ozureprubart (JYB1904), excluding mainland China, Macau, Taiwan, and Hong Kong. GlaxoSmithKline will also be responsible for paying success milestones and royalties for Ozureprubart (JYB1904) to RAPT’s partner, Shanghai Jeyou Pharmaceuticals.
GSK’s substantial investment represents the highest recognition of Jeyou Pharma’s original innovation capabilities and the quality of its clinical data, signaling that the global commercialization process for this drug has been put on the “fast track.”
For Jeyou Pharma, the transition from a biotech company (RAPT) to a multinational corporation (GSK) signifies that JYB1904 has achieved a perfect “strategic upgrade.” This upgrade will directly empower Jeyou Pharma in four key areas, providing the substantial capital needed to advance toward becoming a comprehensive pharmaceutical giant (Big Pharma):
1. Accelerated R&D, with milestone payments expected to be realized earlier: GSK possesses a world-class clinical development system. Following the handover, JYB1904’s late-stage clinical trials in areas such as food allergies will receive more substantial resource support, significantly shortening the time to market and enabling Jeyou Pharma to receive R&D and commercialization milestone payments at each stage sooner.
2. Expansion of Indications, Increasing Milestone Payment Opportunities: Given JYB1904’s broad-spectrum potential in the field of immune inflammation, GSK is highly likely to initiate clinical studies for multiple new indications. Each new indication represents an additional milestone payment milestone, further enhancing the commercial value of this pipeline.
3. Robust Risk Management Capabilities, Significantly Enhancing Clinical Success Rates: New drug development carries extremely high risks. Jeyou Pharma has completed proof-of-concept (POC) studies and provided solid data, while GSK, as a long-established pharmaceutical giant, possesses exceptional expertise in regulatory submissions and risk management, which will greatly ensure the success rate of JYB1904 in subsequent large-scale confirmatory clinical trials and marketing authorization applications.
4. Global marketing network maximizes sales royalties: The peak sales volume after a drug’s market launch directly determines the amount of royalties Jeyou Pharma will receive. GSK possesses a robust global marketing network capable of fully realizing JYB1904’s market potential, thereby generating substantial royalty revenue for Jeyou Pharma.
Industry Insight: The “Value Cycle” of Original Innovation
The success of JYB1904 is by no means an isolated case, but rather the inevitable outcome of Jeyou Pharma’s platform-based R&D capabilities.
As early as the project initiation phase, Jeyou Pharma’s scientific team, guided by forward-looking scientific judgment, identified and optimized this molecule with First-in-Class potential. In 2024, Jeyou Pharma chose to grant global rights to RAPT precisely because of the latter’s development expertise in specific overseas indications, such as food allergies.
Now, with GSK taking the reins, this value loop—“Discovery and Validation in China → Global Strategic Positioning → Commercialization by an MNC”—has been perfectly validated. Looking across the biopharmaceutical sector, there are very few biotech companies that have garnered the favor of two top-tier global multinational pharmaceutical companies (MNCs) simultaneously, yet Jeyou Pharma has achieved this:
From Roche to GSK, and from JMKX002992 to JYB1904, the trend of “both small and large molecules gaining favor” clearly demonstrates that Jeyou Pharma not only possesses the systematic R&D capabilities to consistently produce world-class innovative drugs, but also the business acumen to integrate top-tier global resources and maximize asset value through strategic partnerships.
“Rooted in Shanghai, Reaching Out to the World.” As 2026 begins, from the completion of a nearly 2 billion yuan Series A funding round to the 2.2 billion USD global acquisition driven by our core asset JYB1904, Jeyou Pharma is demonstrating its role as a “pioneer among successful companies” through a series of tangible achievements. We will continue to strengthen our R&D foundation, ensuring that more “Jeyou-developed” products become a source of hope for patients worldwide.
Previous
Validating a Comprehensive R&D System and Pioneering New Frontiers in Combination Therapies | Jeyou Pharma’s First Class 1 Innovative Drug, Jielemei®, Approved for Market Launch
Next
Xu Lei, Member of the Standing Committee of the Pudong New Area Party Committee and Deputy District Governor, Led a Delegation on a Visit to Jeyou Pharmaceutical: Government and Enterprise Join Forces to Chart a Course for Innovation in the Biopharmaceuti

The new indication is chronic spontaneous urticaria; the previously approved treatment for moderate-to-severe allergic asthma has completed Phase I trials……
2024/09/01
Both drugs are Class 1 new drugs, targeting sepsis and IgA nephropathy, respectively……
2024/08/01
The treatment areas include cancer, kidney disease, and infectious diseases...
2023/25/12