Previous
Jeyou Pharma Receives Approval for Clinical Trials of Four Class 1 New Drugs
Shanghai Jeyou Pharmaceutical Technology Co., Ltd. (hereinafter referred to as “Jeyou Pharmaceutical”) announced that JMKX000197 Injection, an innovative small-molecule chemical drug developed by its Small Molecule Innovation Research Institute, has recently been approved by the National Medical Products Administration (NMPA) to proceed with clinical trials for the treatment of patients with malignant pleural effusion.
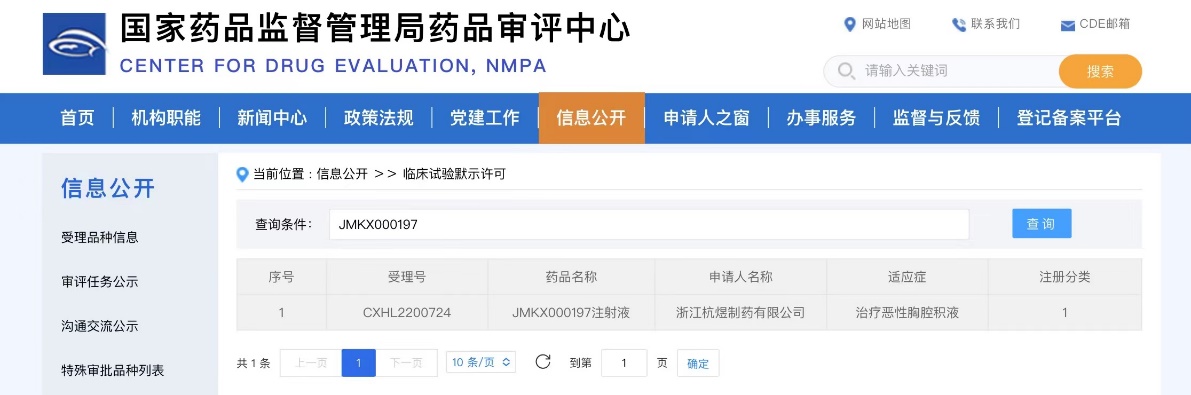
Approximately 40% of malignant pleural effusions (MPE) observed clinically are caused by malignant tumors, with lung cancer, breast cancer, and lymphoma being the primary causes of MPE. Currently, there is a lack of effective treatment options for MPE, resulting in significant unmet clinical needs.
JMKX000197 Injection is a novel small-molecule immune agonist developed by Jeyou Pharmaceuticals. It induces the release of multiple cytokines, triggering an inflammatory response while simultaneously activating macrophages, dendritic cells, and cytotoxic T cells within the body. Through the coordination of these multiple mechanisms, it achieves therapeutic effects.
JMKX000197 Injection activates the body’s immune system. Following local intrathoracic injection, it is rapidly absorbed and cleared. Its systemic circulation concentration is low and of short duration, thereby avoiding the adverse reactions associated with sustained immune system activation. Maintaining high local activity offers excellent clinical prospects for the treatment of MPE. The development of MPE indications based on this target’s mechanism of action represents a global first. Preclinical animal efficacy studies have demonstrated promising results, which will be further validated in clinical trials, with the aim of bringing benefits to patients through innovative therapies.

The new indication is chronic spontaneous urticaria; the previously approved treatment for moderate-to-severe allergic asthma has completed Phase I trials……
2024/09/01
Both drugs are Class 1 new drugs, targeting sepsis and IgA nephropathy, respectively……
2024/08/01
The treatment areas include cancer, kidney disease, and infectious diseases...
2023/25/12