Previous
Jeyou Pharma's Innovative GARP/TGF-β1 Monoclonal Antibody Receives Approval for Clinical Trials
Shanghai Jeyou Pharmaceutical Technology Co., Ltd. announced that innovative drugs developed by its Small Molecule Innovation Research Institute have recently received four Class 1 new drug clinical trial approvals, with three approved by the National Medical Products Administration and one by the U.S. FDA. These approvals cover the fields of kidney disease and cardiovascular and cerebrovascular diseases, demonstrating the company’s innovative R&D capabilities.
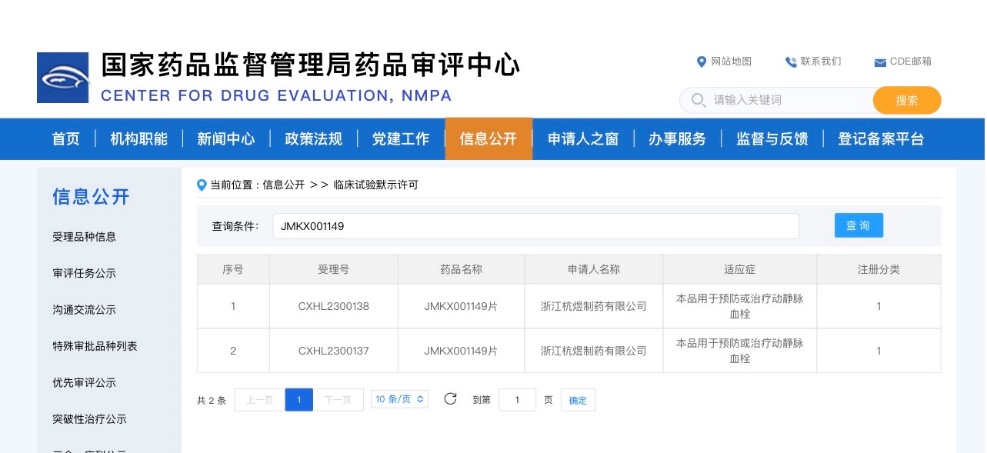
JMKX001149 oral tablets were approved by the National Medical Products Administration on April 17, authorizing the conduct of clinical trials for the prevention and treatment of venous thromboembolism. JMKX001149 is a novel oral anticoagulant indicated for the prevention and treatment of venous and arterial thrombosis. It is particularly effective for postoperative anticoagulation, as well as the treatment and prevention of myocardial infarction and stroke, while simultaneously reducing the risk of bleeding. This drug is expected to meet clinical needs for these chronic conditions and bring benefits to patients.
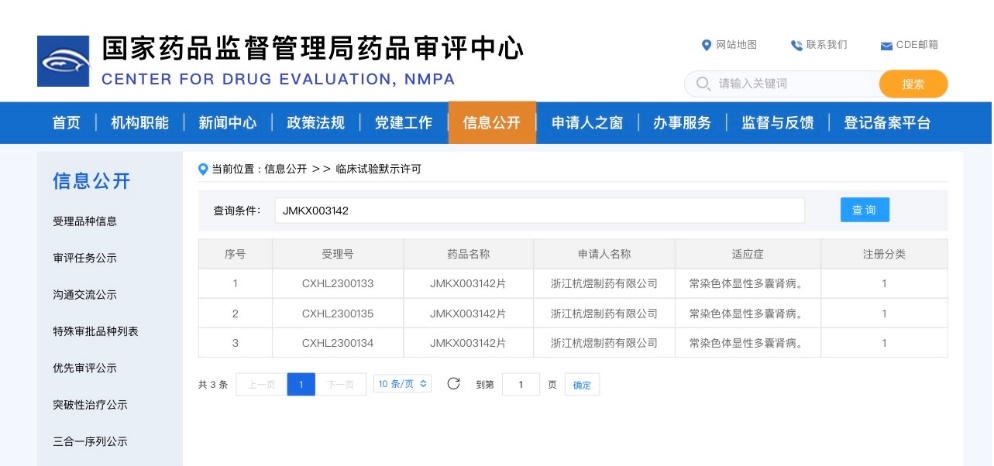
JMKX003142 oral tablets were approved by the National Medical Products Administration on April 17, granting authorization to conduct clinical trials for the treatment of autosomal dominant polycystic kidney disease (ADPKD). There are 1.5 million patients with ADPKD in China; some of these patients progress to end-stage renal disease, requiring dialysis or kidney transplantation, which places a significant burden on their families and society. Currently, there is a lack of targeted therapeutic drugs in China. JMKX003142 is the first new drug of its kind in the country and is expected to meet clinical needs.
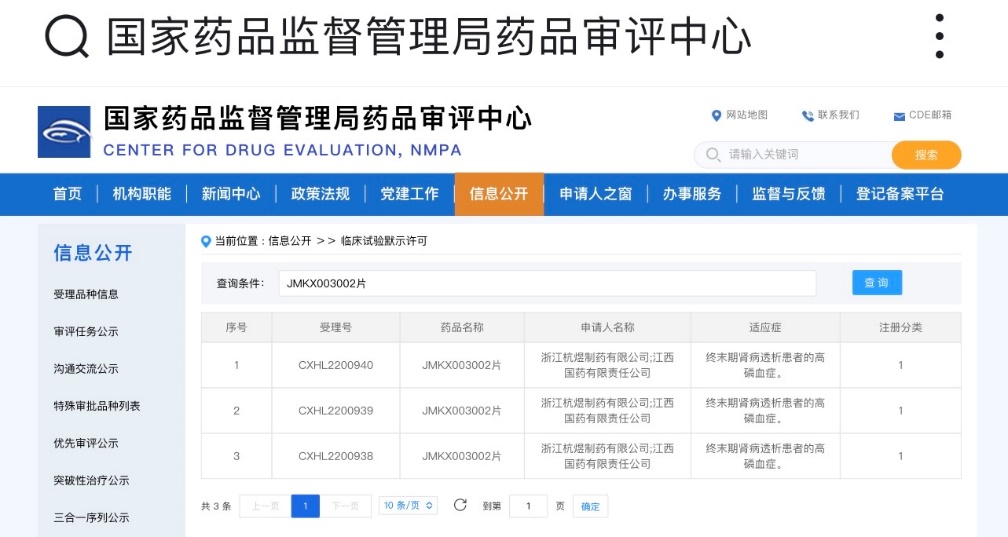
JMKX003002 oral tablets were recently approved by the National Medical Products Administration (NMPA), which has authorized the conduct of clinical trials for the treatment of hyperphosphatemia in patients with end-stage renal disease (ESRD) undergoing dialysis. The total number of people with chronic kidney disease (CKD) in China is estimated to be as high as 120 million. CKD can lead to hyperphosphatemia and associated mineral and bone disorders (CKD-MBD). This product is a latest-generation phosphate binder with good oral compliance. Compared to competing products, it exhibits higher in vitro persistence, offering the potential for sustained reduction of serum phosphorus levels and improvement in the quality of life for patients with kidney disease.

JMKX000189 Injection was recently approved by the U.S. FDA, which has authorized the conduct of clinical trials for the treatment of acute ischemic stroke. China currently has approximately 28 million stroke patients, with about 4 million new cases occurring annually; acute ischemic stroke poses a serious threat to human health. JMKX000189 is the first drug globally to target this specific mechanism for stroke treatment. This novel drug regulates and treats stroke through multiple mechanisms, including modulating immune cells, maintaining vascular integrity, and reducing neuronal apoptosis. Human clinical trials are expected to commence in the near future.

The new indication is chronic spontaneous urticaria; the previously approved treatment for moderate-to-severe allergic asthma has completed Phase I trials……
2024/09/01
Both drugs are Class 1 new drugs, targeting sepsis and IgA nephropathy, respectively……
2024/08/01
The treatment areas include cancer, kidney disease, and infectious diseases...
2023/25/12